 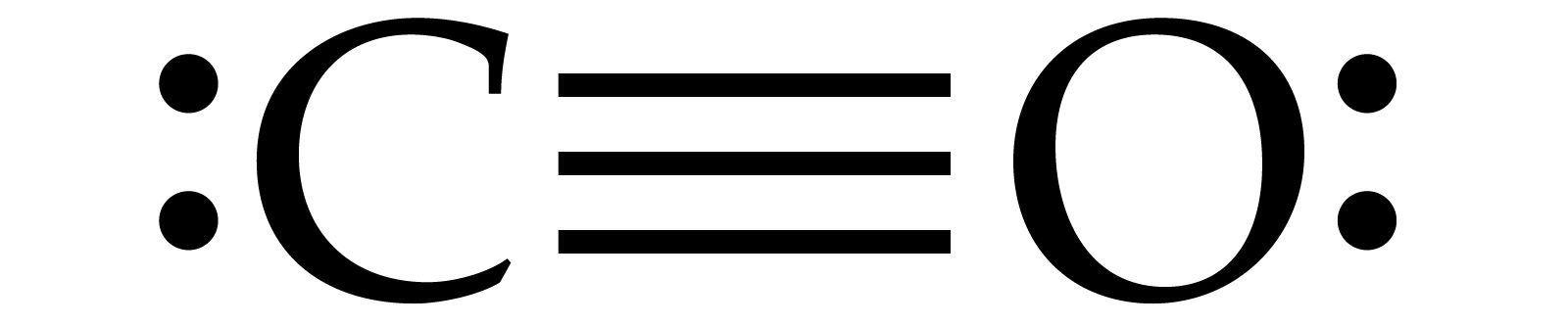
Apart from the toxicity of carbon monoxide, indigenous Native Americans may have experienced the neuroactive properties of carbon monoxide through shamanistic fireside rituals. The early development of metallurgy and smelting technologies emerging circa 6,000 BC through the Bronze Age likewise plagued humankind from carbon monoxide exposure. Early humans probably discovered the toxicity of carbon monoxide poisoning upon introducing fire into their dwellings. Humans have maintained a complex relationship with carbon monoxide since first learning to control fire circa 800,000 BC. It is isoelectronic with cyanide anion CN −. In mammalian physiology, carbon monoxide is a classical example of hormesis where low concentrations serve as an endogenous neurotransmitter ( gasotransmitter) and high concentrations are toxic resulting in carbon monoxide poisoning. It is produced by many organisms, including humans. Ĭarbon monoxide has important biological roles across phylogenetic kingdoms. Upon emission into the atmosphere, carbon monoxide affects several processes that contribute to climate change. In industry, carbon monoxide is important in the production of many compounds, including drugs, fragrances, and fuels. Numerous environmental and biological sources generate carbon monoxide. The most common source of carbon monoxide is the partial combustion of carbon-containing compounds. 
It is a key ingredient in many processes in industrial chemistry. In coordination complexes, the carbon monoxide ligand is called carbonyl. 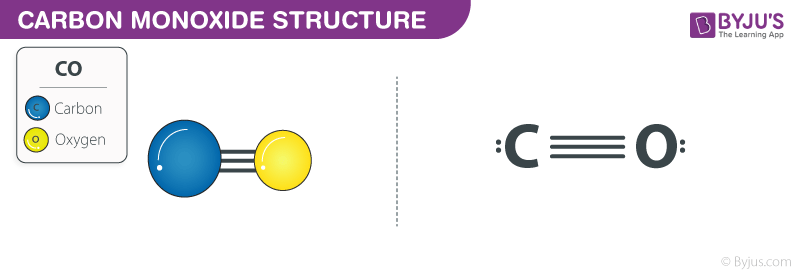
Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. I hope you have completely understood all the above steps.įor more practice and better understanding, you can try other lewis structures listed below.Carbon monoxide ( chemical formula CO) is a poisonous, flammable gas that is colorless, odorless, tasteless, and slightly less dense than air. By doing so, you will get the following lewis structure of CO. In the above lewis dot structure of CO, you can also represent each bonding electron pair (:) as a single bond (|). You can see from the above picture that the carbon atom as well as oxygen atom both are forming an octet.Īnd hence the above lewis dot structure of CO molecule is stable. 
So again we have to shift one more electron pair from the oxygen atom.Īfter shifting this electron pair, the carbon atom will get 2 more electrons and thus its total electrons will become 8. Now to make this carbon atom stable, you have to shift the electron pair from the oxygen atom.īut after shifting one electron pair, the carbon atom is still not forming an octet as it has only 6 electrons. Carbon has only 4 electrons and it is unstable. Unfortunately, this carbon atom is not forming an octet here. In order to check the stability of this carbon (C) atom, we have to check whether it is forming an octet or not. In this step, you have to check whether the central carbon atom (C) is stable or not. If it does not have octet, then shift the lone pair to form a double bond or triple bond. Step 5: Check the octet on the central atom. Total valence electrons in CO molecule = valence electrons given by 1 carbon atom + valence electrons given by 1 oxygen atom = 4 + 6 = 10. You can see the 6 valence electrons present in the oxygen atom as shown in the above image. Hence the valence electrons present in oxygen is 6. Oxygen is group 16 element on the periodic table. → Valence electrons given by oxygen atom: You can see the 4 valence electrons present in the carbon atom as shown in the above image. Hence the valence electrons present in carbon is 4. → Valence electrons given by carbon atom:Ĭarbon is group 14 element on the periodic table. Here, I’ll tell you how you can easily find the valence electrons of carbon as well as oxygen using a periodic table. (Valence electrons are the electrons that are present in the outermost orbit of any atom.) In order to find the total valence electrons in CO (carbon monoxide) molecule, first of all you should know the valence electrons present in a single carbon atom as well as oxygen atom. Steps of drawing CO lewis structure Step 1: Find the total valence electrons in CO molecule 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
