As those surface layers gradually mix into deep water, the entire ocean is affected. 
(Scientists call this stabilizing effect “buffering.”) But so much carbon dioxide is dissolving into the ocean so quickly that this natural buffering hasn’t been able to keep up, resulting in relatively rapidly dropping pH in surface waters. Scientists formerly didn’t worry about this process because they always assumed that rivers carried enough dissolved chemicals from rocks to the ocean to keep the ocean’s pH stable. In the past 200 years alone, ocean water has become 30 percent more acidic-faster than any known change in ocean chemistry in the last 50 million years. Even though the ocean is immense, enough carbon dioxide can have a major impact. When carbon dioxide dissolves in seawater, the water becomes more acidic and the ocean’s pH (a measure of how acidic or basic the ocean is) drops. But in the past decade, they’ve realized that this slowed warming has come at the cost of changing the ocean’s chemistry. Since the beginning of the industrial era, the ocean has absorbed some 525 billion tons of CO 2 from the atmosphere, presently around 22 million tons per day.Īt first, scientists thought that this might be a good thing because it leaves less carbon dioxide in the air to warm the planet. At least one-quarter of the carbon dioxide (CO 2) released by burning coal, oil and gas doesn't stay in the air, but instead dissolves into the ocean. 
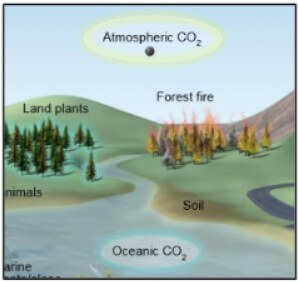
Burning fossil fuels in cars or power plants is another way this carbon can be released into the atmospheric reservoir quickly.Ocean acidification is sometimes called “climate change’s equally evil twin,” and for good reason: it's a significant and harmful consequence of excess carbon dioxide in the atmosphere that we don't see or feel because its effects are happening underwater. Processes such as erosion release this carbon back into the atmosphere very slowly, while volcanic activity can release it very quickly. When these organisms died, slow geologic processes trapped their carbon and transformed it into these natural resources. Rocks like limestone and fossil fuels like coal and oil are storage reservoirs that contain carbon from plants and animals that lived millions of years ago. Two-way carbon exchange can occur quickly between the ocean’s surface waters and the atmosphere, but carbon may be stored for centuries at the deepest ocean depths. The ocean plays a critical role in carbon storage, as it holds about 50 times more carbon than the atmosphere. Respiration, excretion, and decomposition release the carbon back into the atmosphere or soil, continuing the cycle. Animals that eat plants digest the sugar molecules to get energy for their bodies. They use energy from the sun to chemically combine carbon dioxide with hydrogen and oxygen from water to create sugar molecules. For example, in the food chain, plants move carbon from the atmosphere into the biosphere through photosynthesis.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
